Anti-Rituximab Antibodies
-
Bioanalytical Antibodies
-
Antibodies for Bioanalysis and Drug Monitoring
-
Our Anti-Biotherapeutic Antibodies
- Anti-Abatacept Antibodies
- Anti-Adalimumab Antibodies
- Anti-Alemtuzumab Antibodies
- Anti-Atezolizumab Antibodies
- Anti-Avelumab Antibodies
- Anti-Belatacept Antibodies
- Anti-Bevacizumab Antibodies
- Anti-Brentuximab Antibodies
- Anti-Cemiplimab Antibodies
- Anti-Certolizumab Antibodies
- Anti-Cetuximab Antibodies
- Anti-Daratumumab Antibodies
- Anti-Denosumab Antibodies
- Anti-Dupilumab Antibodies
- Anti-Durvalumab Antibodies
- Anti-Eculizumab Antibodies
- Anti-Etanercept Antibodies
- Anti-Evolocumab Antibodies
- Anti-Golimumab Antibodies
- Anti-Infliximab Antibodies
- Anti-Ipilimumab Antibodies
- Anti-Natalizumab Antibodies
- Anti-Nivolumab Antibodies
- Anti-Obinutuzumab Antibodies
- Anti-Ocrelizumab Antibodies
- Anti-Omalizumab Antibodies
- Anti-Palivizumab Antibodies
- Anti-Panitumumab Antibodies
- Anti-Pembrolizumab Antibodies
- Anti-Pertuzumab Antibodies
- Anti-Ranibizumab Antibodies
- Anti-Rituximab Antibodies
- Anti-Secukinumab Antibodies
- Anti-Tocilizumab Antibodies
- Anti-Trastuzumab Antibodies
- Anti-Ustekinumab Antibodies
- Anti-Vedolizumab Antibodies
-
Our Anti-Biotherapeutic Antibodies
-
Antibodies for Bioanalysis and Drug Monitoring
s
s
s
Antibodies for bioanalysis and drug monitoring of rituximab and biosimilars
Develop highly selective and sensitive PK and ADA assays for rituximab (Rituxan, MabThera) using our range of ready-made anti-idiotypic antibodies.
- Recombinant inhibitory antibodies specific to rituximab
- Rat anti-rituximab clone MB2 A4 for ELISA and flow cytometry
- Fully human surrogate positive control or calibrator
- Sequence defined, well characterized reagents with secure supply
- Recombinant production, stringent quality control for batch to batch consistency
- Non-animal-derived antibodies, reducing the use of animals in science
Table 1. Antibodies Specific to Rituximab.
Specificity |
Catalog Number |
Clone |
Format |
Affinity* KD, nM |
Assay Recommendation |
Product Details |
|---|---|---|---|---|---|---|
|
Rituximab Inhibitory Type 1 |
MCA2260 |
MB2 A4 |
Rat IgG2a HRP FITC |
N/A |
PK bridging ELISA Flow cytometry |
|
|
HCA061 |
AbD02842 |
Fab-dHLX-MH1 HRP |
4.9 |
PK bridging ELISA |
||
|
HCA062 |
AbD02844 |
Fab-dHLX-MH1 HRP |
10.0 |
PK bridging ELISA |
||
|
HCA186 |
AbD18423 |
Fab-FH2 |
0.13 |
PK bridging ELISA |
||
|
HCA201 |
AbD18423_hIgG1 |
hIgG1 |
0.13 |
PK bridging ELISA ADA control |
* Affinity measured in the monovalent Fab format
1 Bivalent Fab antibody, double Helix Loop Helix motif dimerization domain, Myc- and His-6-tags
2 Monovalent Fab antibody DYKDDDDK- and His-6-tags
Anti-Rituximab Inhibitory Antibodies (Type 1)
Type 1 anti-rituximab antibodies inhibit the binding of the drug rituximab to its target, the B cell surface marker CD20. They are ideal for development of a pharmacokinetic (PK) bridging ELISA to measure free drug, and for flow cytometry assays. The antibody in fully human IgG1 format is suitable as a surrogate positive control or calibrator for an anti-drug antibody (ADA) assay.
ELISA Protocols to Get You Started
PK Bridging ELISA
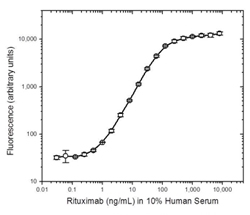
Fig. 1. Rituximab PK bridging ELISA using antibodies HCA186 and MCA2260P.
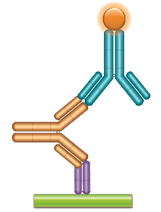
Schematic image of PK bridging ELISA measuring free drug. Anti-idiotypic capture antibody, Fab format (purple), monoclonal antibody drug (gold), anti-idiotypic detection antibody, Ig format (blue), labeled with HRP.
ADA Bridging ELISA
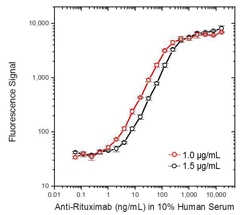
Fig. 2. Rituximab ADA bridging ELISA using antibody HCA201.
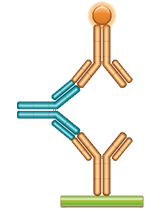
Schematic image of ADA bridging assay. Monoclonal antibody drug as capture antibody and detection antibody labeled with HRP (gold), fully human anti-idiotypic antibody, Ig format (blue).
Ocrelizumab
Ocrelizumab is a second-generation humanized anti-CD20 antibody that binds to an epitope on CD20 that overlaps with the epitope to which rituximab binds (Klein et al. 2013). Rat Anti-Rituximab Antibody, clone MB2A4 (MCA2260) recognises both rituximab and ocrelizumab in a specificity titration ELISA.
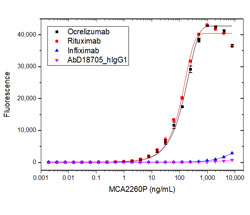
Fig. 3. Specificity titration ELISA using MCA2260P.
Related Products
Reference
- Klein C et al. (2013). Epitope interactions of monoclonal antibodies targeting CD20 and their relationship to functional properties. mAbs 5:22–33.
Resources
Licensed Use: For in vitro research purposes and for commercial applications for the provision of in vitro testing services to support preclinical and clinical drug development. Any re-sale in any form or any other commercial application needs a written agreement with Bio-Rad.